For people with paralysis, losing the ability to speak can be just as devastating as losing the ability to move. You may know exactly what you want to say, but your body can’t turn those thoughts into words. A US startup called Paradromics just took a major step toward changing that.
The US Food and Drug Administration (FDA) has approved a small human trial of a fully implantable brain-computer interface (BCI) designed to help people communicate again using only their brain signals.
What this brain chip actually does
This device does not read minds. It doesn’t create thoughts or control behavior. Instead, it listens.
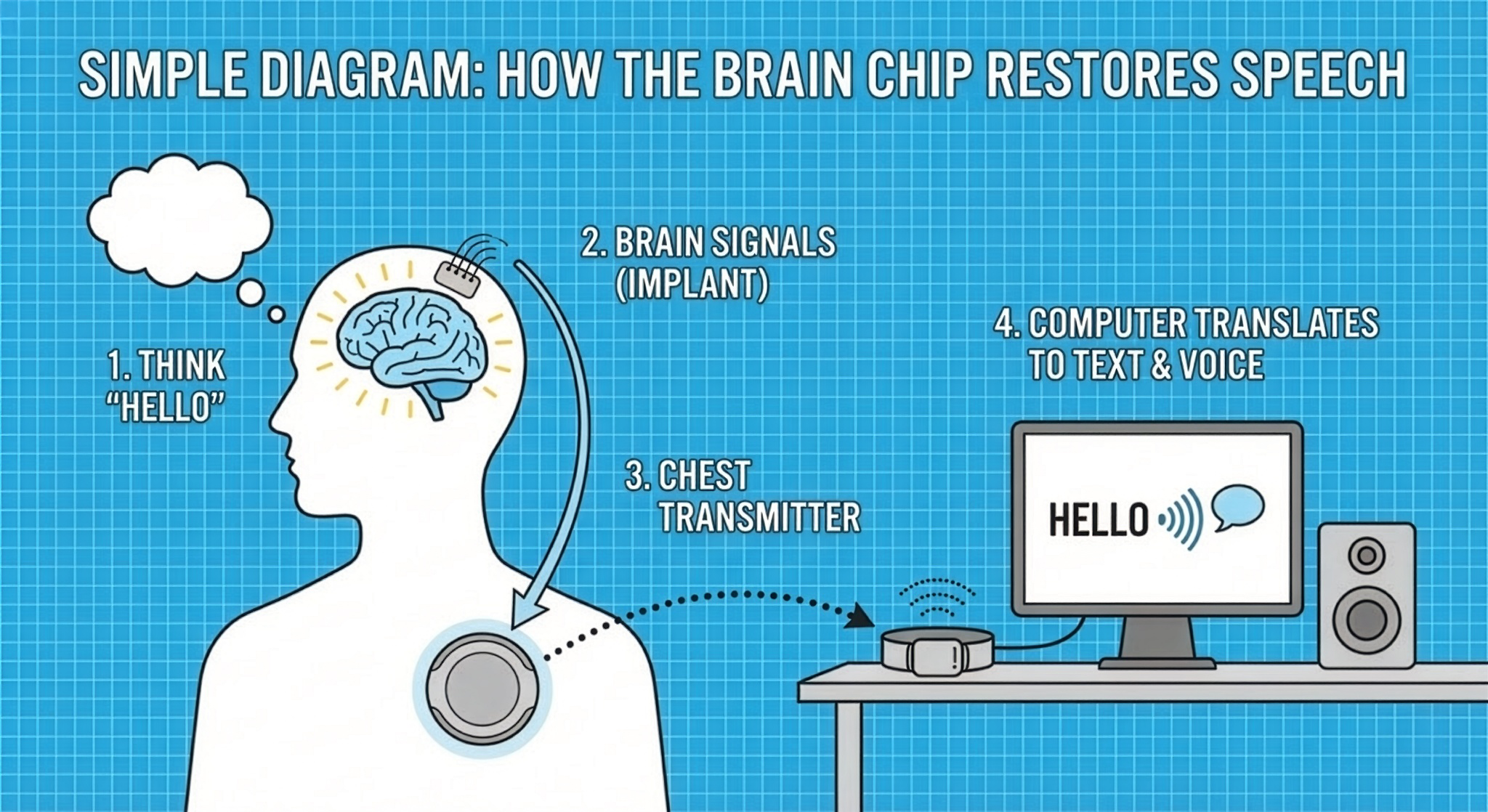
When you think about speaking, specific neurons in your brain fire in recognizable patterns. The Paradromics device is designed to detect those tiny electrical signals and send them to a computer, where software translates them into text on a screen or a synthetic voice.
In simple terms: you think about speaking, and the system tries to turn that intention into words.
How the device works
The system is made of several parts that work together:
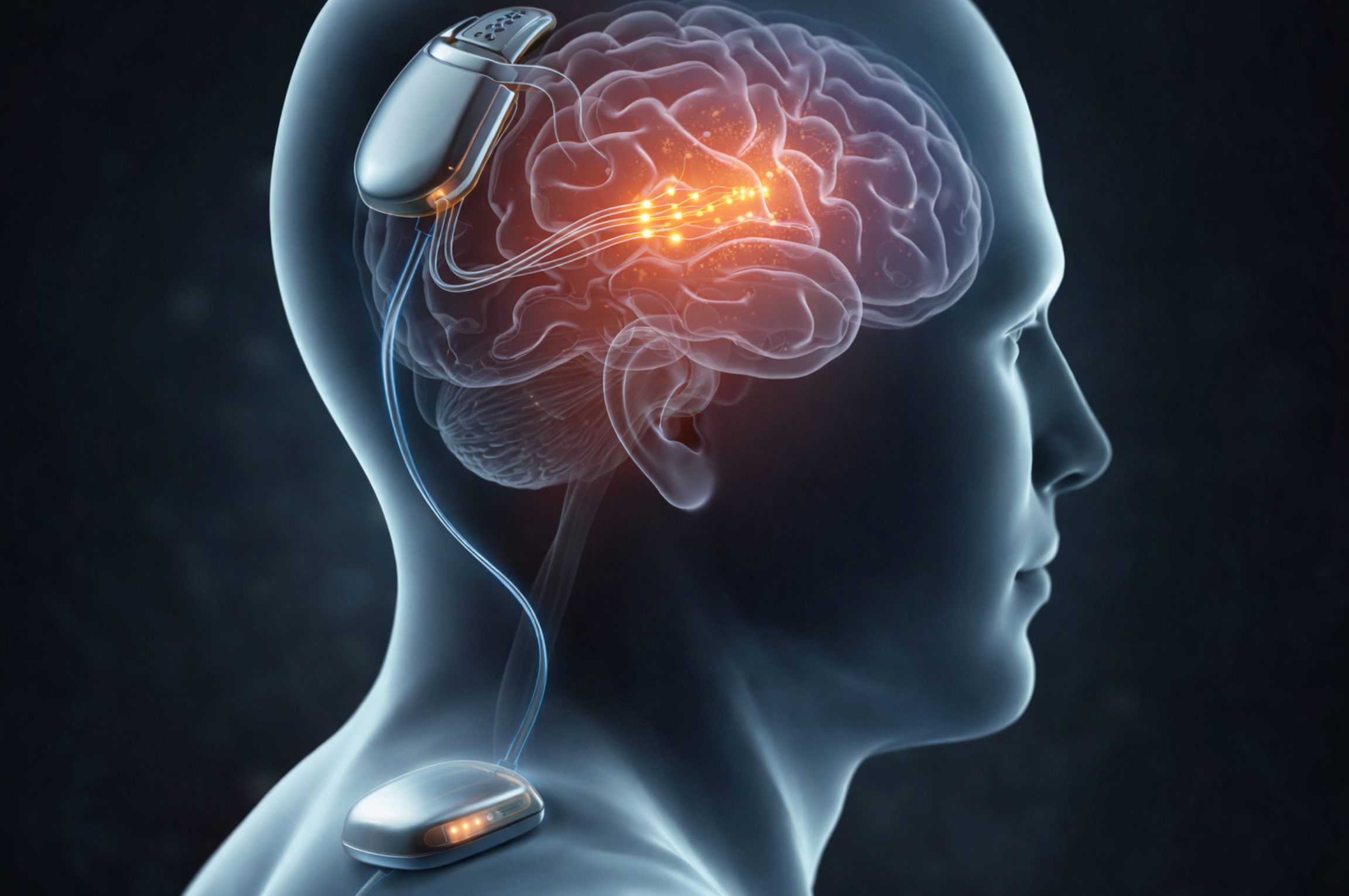
• A small titanium device implanted under the skin
• Over 400 ultra-thin electrodes placed just beneath the surface of the brain
• A wireless transmitter implanted in the chest
• An external wearable transmitter and a small computer
The electrodes sit near neurons in the part of the brain that controls the lips, tongue, and voice box. Each electrode is thinner than a human hair. These electrodes pick up brain signals and send them through a thin cable under the skin to the chest transmitter.
From there, the data is sent wirelessly to an external device, which powers the implant and passes the information to software that analyzes what the person is trying to say.
What the human trial looks like
The trial will start very small. Only two people will participate at first.
Each participant will receive one implant placed about 1.5 millimeters into the brain. They will be shown sentences and asked to imagine speaking them. Over time, the system learns which brain signals match which sounds or words.
The goal is real-time communication using text or a synthesized voice, potentially based on recordings of how the person sounded before they lost speech.
Researchers will also test whether the device can detect imagined hand movements, which could allow users to control a computer cursor.
Why some people are excited
For many people with paralysis, even temporary communication would be life-changing.
Some researchers compare these trials to early space missions: dangerous, limited, and temporary, but necessary to move forward. Over the last two decades, brain implants have gone from working for weeks to working for years.
This trial is also the first FDA-approved study focused specifically on synthetic speech generation using a fully implantable BCI.
The biggest concern: rejection and scar tissue
Not everyone is convinced this technology can work long term.
One of the biggest problems with brain implants is the body’s immune response. Over time, brain tissue can form scar tissue around electrodes. This doesn’t “reject” the implant like a bad organ transplant, but it can weaken or block the signals.
Some former patients with other brain implants have seen benefits fade after months or years. Engineers have tried flexible wires, stiff wires, coatings, and ultra-small electrodes, with mixed results.
Critics argue that giving people hope without long-term solutions can feel cruel. Supporters respond that trial participants fully understand the risks and that modern medicine only exists because people were willing to take those risks.
Is this a cure?
No. This is not a cure.
Even researchers involved are clear about that. The goal is mitigation, not reversal. The hope is to give people a way to communicate again, even if it doesn’t last forever.
Other approaches, including non-surgical brain stimulation and stem cell research, are also being explored. This trial is just one path among many.
Why this still matters
The FDA approval doesn’t mean success is guaranteed. It means the technology has reached a point where it’s safe enough to test in humans under strict oversight.
If it works even for a limited time, it could dramatically improve quality of life for people who currently have no way to communicate.
This is not the end of the story. It’s an early chapter.






